When many of us think of helium, we tend to think of a “funny” gas used to blow up party balloons and make our friends sound like chipmunks! However, helium is a very powerful gas that shouldn’t be played with irresponsibly! Here are some fun facts about helium you’ll want to remember.
1. It is represented by the symbol “He.”
On the periodic table, helium is represented by the chemical symbol “He”. Helium is known for being the second lightest and also the second most abundant element in the known universe, just after hydrogen. It has the atomic number 2, funnily enough, just behind hydrogen with an atomic number of 1!
2. It was discovered by Pierre Janssen.
Helium was first said to have been spotted by the French astronomer Pierre Janssen while he observed an eclipse of the sun back in 1868. Sir William Ramsay, a chemist from Scotland, later discovered the element himself in 1895.
3. It is lighter than air.
Although we don’t tend to think of air as being something particularly weighty, helium is even lighter! That is why helium is so useful for filling balloons and airships.

4. It is a noble gas.
Helium is considered to be a noble gas due to its chemical properties. That is to say that it is colourless, odourless, and does not readily react with other elements.
5. Helium is rarely found in nature.
Although helium is the known as the second most abundant element in our universe, it is actually relatively rare here on Earth! Most of Earth’s helium comes from the radioactive decaying of heavier elements like thorium.
6. It has a very low boiling point.
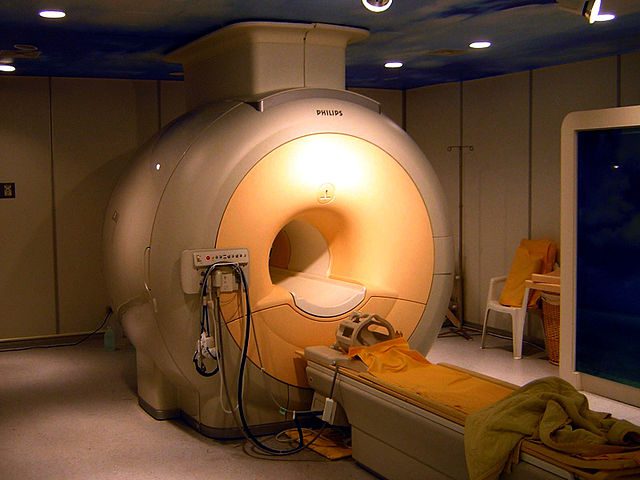
Helium is famous for having the lowest boiling point of any element! It becomes a liquid at temperatures close to absolute zero (-269 degrees Celsius or -452 degrees Fahrenheit). That is why it is essential for cooling superconducting magnets in MRI machines and particle accelerators.
7. It can alter your voice!
One of the most famous things about helium is that it can temporarily change the pitch of a person’s voice. This happens because helium is less dense than air, causing sound to travel faster through it.
8. Helium is an essential component in deep-sea diving equipment.
Helium is used in deep-sea diving breathing mixtures. When combined with oxygen, it can help prevent nitrogen narcosis, a potentially dangerous condition caused by breathing high-pressure nitrogen. This is because helium has low solubility in water, which makes it less likely to dissolve in the bloodstream compared to nitrogen. This is important for avoiding decompression sickness in deep-sea diving.
9. It’s useful in space exploration, too – particular during the Apollo missions.
Liquefied helium is used to cool and liquefy hydrogen in rocket engines. It played a crucial role in the Apollo missions to the Moon – it certainly helped to get Armstrong, Aldrin, and Collins back safely!
10. There is an ongoing helium shortage
There are great concerns about the global helium shortage we are currently dealing with. The shortage is largely due to the limited supply and increased demand for various scientific, medical, and industrial applications.
11. Helium-3 may become essential to us.
Helium-3 is a very rare isotope of helium with one less neutron than the more common helium-4 isotope. It has potential applications in nuclear fusion research and as a fuel for future spacecrafts!

FAQs about Helium
What is helium used for?
Helium is commonly used by tires, refrigerators, fire extinguishers, aerosol products, and air conditioner manufacturers. They use the element to test seals before their products can be sold. Helium is also commonly used in deep-sea diving and space science. It’s also frequently used to help inflate balloons and large aircraft, as it can float lighter than air.
Why is helium a special element?
Helium, as a noble gas, is considered to be special as it is unique. It has a very low boiling point, low solubility, low density, and high thermal conductivity. Therefore, it has a myriad of uses.
Can we create helium?
No, there is no chemical way for us to create helium. That is one of the reasons we are fast running out of it – despite it being abundant across the universe.
Further reading
https://www.rsc.org/periodic-table/element/2/helium
https://education.jlab.org/itselemental/ele002.html
Do you know any interesting facts about Helium? Share them in the comments below!